Aggressive modern enzymes require review of diet formulations
There are considerable opportunities available for co-operative use of xylanase and phytase in poultry diets. However, conservative modification to digestible phosphorous and energy concentrations are increasingly obsolete as new products elicit profound changes on how the bird perceives what it ingests.
By Aaron Cowieson, AB Vista Feed Ingredients, UK
From relatively humble beginnings in wheat- and barley-based diets for broilers, the global feed enzyme market is today worth over US$550 mln, and saves the global feed market an estimated $3-5 bln per year. The rapid growth of this market, notably in the past 5-10 years, is associated primarily with the uptake of phytase technology and the wider use of carbohydrases in corn-based diets, both of which are in response to increasing input costs. Considerable investment in application research has led to a number of strategic developments in the use of exogenous enzymes, including a greater understanding of calcium and sodium matrices for phytases, and the extent of cooperativity between phytases and carbohydrases.Historically phytases have been used rather ‘crudely’ as a cost-effective replacement for sources of inorganic phosphorous, but recent evidence suggests that liberation of orthophosphate from phytin may be of secondary importance to the removal of reactive phytate from the ration. Radical diet modification is required to formulate in the absence of phytate, and these changes extend to calcium, sodium, amino acids and energy.
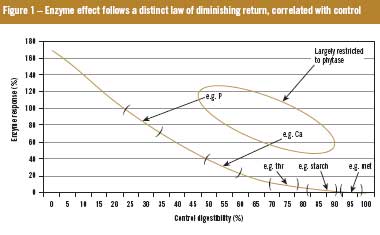
Pivotally, it has also recently been demonstrated that the magnitude of effect of exogenous xylanases depends on the nutritional value of the diet to which they are added, precluding the possibility of full additivity between xylanase and phytase (Figure 1). Thus, in the future, more important than characteristics of the enzyme product may be the application know-how to extract maximum return from its use. The strategic use of phytases and xylanases with appropriate diet modification can result in considerable economic advantages for the end user, but these advantages may be blunted if matrix values are mis-assigned.
Accommodation of enzyme effect
In many ways the primary function of nutrition research – at least for the past few decades – has been to keep up with changing genetic potential offered to nutritionists by the animal breeding companies. Thus, nutrition research has been reactive rather than pro-active in nature where the new breeds arrive on the market and nutritionists must respond with (usually modest) changes to nutrient requirements.
Initially, exogenous enzymes slotted into this conservative and ‘time honoured’ environment with relative ease and small (but nonetheless valuable) dietary modifications ensued. An example of this is the incorporation of xylanase into a corn/soy-based broiler diet, and the removal of a small percentage of the added fat (perhaps 30-60 kcal/kg equivalent) in the grower or finisher phase. The net result was a saving of several US dollars per tonne of feed, and often an improvement in flock performance and uniformity.However, phytase does not fit as readily into a conservative feed formulation tradition. Accommodation of its considerable and wide-ranging effect by modest adjustments to (only) inorganic phosphate concentrations may create more problems than it solves. Phytase, particularly the 2nd and 3rd generation bacterial phytases, are engineered in such a way that they are extremely aggressive at liberating orthophosphate and of removing, almost completely, the anti-nutritive effects of phytate in the process. Whilst the liberated phosphorus can be quantified with relative ease, the extra-phosphoric effects of phytases are much more difficult to contend with. These extra-phosphoric effects must be considered to be a potentially valuable up-side to the use of phytase, but at the same time a considerable threat to productivity, litter/footpad quality and bone strength if not appropriately accounted for in diet formulation.
Extra-phosphoric effects
Poultry breeding regimes have not, at least historically, taken into consideration the influential nature of phytate. Phytate is a ubiquitous (Table 1), reactive and extremely influential compound with physiological effects far beyond digestible phosphorus dilution. In feed, phytate is found at concentrations of between 7-20 g/kg (~0.2-0.5 g/kg phytate-P) and is usually present as an unreactive salt of magnesium and potassium (sometimes calcium is also implicated) known as ‘phytin’. Once ingested and exposed to acidic conditions H+ ions readily replace the divalent cations and phytate is solubilised as an almost fully protonated moiety.
It is the reduction in protein solubility which is responsible for the compromising effect of phytate on sodium, calcium, amino acids and energy as hyper-secretion of HCl, mucin, pepsin, bile and NaHCO3 results, increasing energy, sodium and amino acid flow in the lumen and interfering with active transport. Additionally, though phytate-protein complexes will be almost entirely recovered by the time the feed reaches the terminal ileum, intact phytate, which reaches the duodenum, will rapidly scavenge divalent cations, especially calcium, forming insoluble precipitates, effectively removing these valuable ions from the digestive milieu. Thus, the ingestion of modest concentrations of phytate (<1% of the diet) has a profound impact on endogenous processes and the solubility and digestibility of dietary nutrients.
Mineral chelation
It is fascinating to consider that nutrient requirements of poultry were set in the presence of phytate (i.e. the absence of phytase) and so, inadvertently, accommodated these consequences. One example of this is the relatively high concentrations of sodium and calcium that are often fed to modern broilers. Of course not all the birds’ sodium and calcium requirements are dictated by phytate, but a substantial (perhaps 30% of the calcium requirement and 15% of the sodium requirement) are. It is no coincidence that sodium is capable of disrupting phytate-protein complexes and so it is likely that ‘pre-phytase’ sodium requirements inadvertently took this mechanism into account. Furthermore, one phytate molecule is capable of complexing with up to 5 molecules of calcium. In a phytate-free ration the associated calcium requirement is only approx. 0.6%. However, it is also important to consider that not all esters of phytate are similarly malignant and so it is not necessary to reduce all phytate to myo-inositol and free phosphate to remove the anti-nutritive effects. There is a disproportionate decrease in the capacity of phytate to bind calcium as phosphates are lost.The new generation bacterial phytases resist proteolytic digestion more than their fungal counterparts and they are very aggressive ‘phytate removers’. The new generation phytases may more appropriately be assigned calcium matrices of 0.2% or even greater, thereby giving rise to both remarkable commercial opportunity and increased negative consequence of misuse.
Formulating
In order to maximise the value of phytases and to mitigate risk of misuse, appropriate modifications to diet nutrient density are required and should include a matrix for phospherous, calcium, sodium, amino acids and energy. In the interests of establishing a frame of reference, it is suggested that for the new 3rdgeneration (engineered E. Coli) phytases appropriate 500 FTU/kg release values could be 0.13-0.14% phosperous, 0.17-0.20% calcium, 0.02-0.05% sodium, 1-6% for amino acids (1% for methionine, 6% for cysteine, with others following an established pattern) and around 40 kcal/kg for ME.
Failure to account for these effects (e.g. application of phosphorous matrices but no associated calcium value) can lead to water retention issues, perhaps mediated via renal damage associated with inappropriately high circulating calcium concentrations.
It has recently been reported that the beneficial effects of exogenous xylanase in poultry and swine diets are inextricably linked to the size of the undigested portion of fat, protein and starch that leaves the ileum. This observation, supported by some 19 peer-reviewed papers published between 1998 and 2009 rules out, by definition, full additivity between pro-nutrients. As phytase (whether credited or not) improves ileal protein, fat and starch digestibility, it reduces the undigested fraction. Figure 2 gives an estimate of this effect. In this situation energy matrices for xylanase should be reduced (by about 20%) in the presence of phytase to acknowledge the now reduced undigested fraction. By definition then, only the first additive of choice can carry its full matrix when added to a ‘virgin’ diet, but subsequent additives should have their martrices discounted to accommodate the influence of the current incumbents.
As theoretical (if not realistic) maximum ileal digestibility is 100%, digestibility enhancing pro-nutrients constantly move digestibility toward that fixed asymptote so opportunity for further improvement declines with each new addition.
Antibiotic growth promoters, coccidiostats and other commonly-used additives in the matrix should be discounted proportionate to the benefits of the incumbents if the matrix values a supplier promotes were established in diets that do not contain phytase. For example, an energy matrix of 100 kcal/kg for xylanase may end up being 50-60 kcal/kg in a diet that contains an array of performance and digestibility-enhancing therapeutics and enzymes.
Conclusion
Formulating in the absence of phytate is not easy and requires appropriate attention to dietary sodium, calcium, amino acids, energy and phosphorous concentrations. However, removal of phytate through the use of the new generation phytases presents unrivalled commercial opportunity to optimise total calcium and sodium concentrations, improving protein digestibility as well as economics associated with diet density, and of course digestibility of phosphorous.As nutrition has taken a reactive role in recent years it is timely that a nutrition-driven development has forced breeders to take note that their products may now not be in line with current feeding practices. Absence of phytate should certainly be a consideration for selection pressure in the future if new breeds are to be harmonised with a new era of phytate-free nutrition.











