Strengthening gut health with macroalgal extracts

Gut health has become increasingly important in the livestock industry with the emergence of antimicrobial resistance and the urge to limit the use of antibiotics. Macroalgal extracts can play an important role within the reduction of antibiotics use in farms.
Part of the Gut Health 2022 Special
In modern production systems, the gastrointestinal tract is being challenged and the subtle balance gut health relies on can be impaired. The components that define gut barrier and immune function can be weakened and lead to higher occurrence of digestive troubles associated with dysbiosis. This disbalance will trigger local and systemic inflammation, affecting the global health status and the growth performance of the animals. The challenge in the industry is to find solutions capable of supporting the epithelial barrier function and the gut-associated lymphoid tissue (GALT) in order to maintain a proper gut health and thus ensure good performance without the need of antibiotics. Recent scientific research and trials in commercial conditions have indicated the potential of in-feed marine macroalgal polysaccharides as reliable agents for gut integrity promotion and immune modulation in young animals. For this, macroalgal extracts can be directly used in the young animals feed and/or indirectly by adding these macroalgal bioactive compounds to maternal feeds as they prove to increase the transfer of immune components and ultimately to improve the health and performance outcome of the progeny.
Figure 1 – Macroalgal polysaccharide structure responsible of their bioactivities. From left to right: branched structure, sugar unit diversity, presence of rare sugars and presence of sulphate groups.
Unique structural features
Parietal polysaccharides of seaweeds present structural complexity and a unique composition that confer them high reactivity and explain their biological properties when used in animals. The complexity and biological reactivity of seaweed polysaccharides stem from the nature of the sugar units, which are diverse and sometimes rare, such as uronic acids, xylose and rhamnose; the variety of glycosidic bonds leading to their branched structure and the presence of sulphate groups (Figure 1). Furthermore, their polyanionic structure and solubility increase their reactivity and enable their recognition by host cells. Sulphated polysaccharides are characteristic of macroalgae (they are not found in terrestrial plants, nor freshwater microalgae nor yeast cell walls). Olmix Group marine bioactive ingredient extraction know-how has led to the development of an in-feed product, Algimun, which is based on the combination of two bioactive macroalgal extracts: MSP Barrier, a red algal extract, which enhances the gut barrier function; and MSP Immunity, a green algal extract, that modulates innate and adaptive immune responses. Algimun bioactive molecules proved to be resistant to feed processing, especially to heat treatment and extrusion.
Gut health promoting properties
The effects of MSP Barrier on intestinal barrier function were demonstrated in an in vivo scientific study. MSP Barrier reduced the paracellular passage of FITC-dextran (gut permeability biomarker) to the blood stream in an animal model known to trigger inflammation in the gut mucosa and consequently a loss of its integrity. Other studies have proven, in vitro, that MSP Barrier upregulates the expression of genes encoding transmembrane and scaffolding proteins which are required for an optimal functioning of the tight junction complexes; and the expression of genes encoding for major mucus proteins which prevents pathogen attachment and colonisation of the gut epithelium.
Figure 2 – TEER of IPEC-1 cell line incubated with E. coli K88 1305 and/or MSP Barrier (kΩ/cm²).
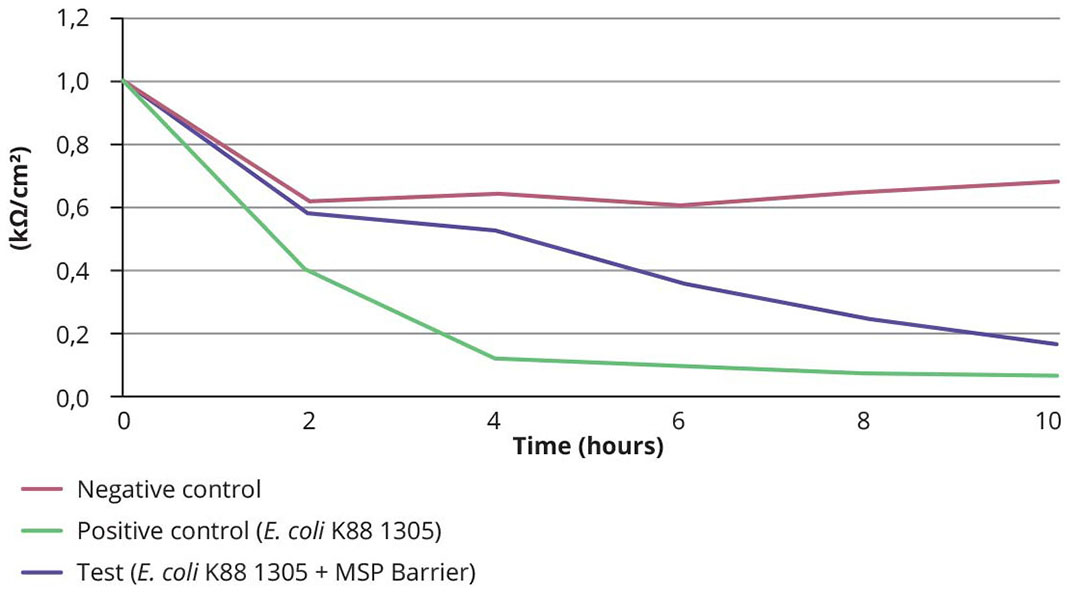
New scientific evidence has corroborated the capacity of MSP Barrier to strengthen the intestinal mucosa. In the first assay, the ability of MSP Barrier to reinforce gut integrity in the presence of a pathogenic strain of Escherichia coli was assessed. IPEC-1 cultures were incubated in the culture medium only (negative control), with E. coli K88 1305 (1.35×108 CFU/mL) (positive control) or with E. coli K88 1305 and MSP Barrier (test). Results showed that the transepithelial electrical resistance (TEER) which is a marker of the epithelium integrity was stable in the negative control (non-infected) in the first 10 hours of monitoring, while it rapidly decreased (within 4 hours) in the positive control (infected). When the IPEC-1 cell line was in contact with MSP Barrier prior to be inoculated with E. coli K88 1305, TEER was maintained at a higher level than in the positive control in the first 10 hours post-infection (Figure 2). In the second assay, IPEC-1 cells were pre-treated with the culture medium only (negative control) or with MSP Barrier at three concentrations (tests: 1, 1/10 and 1/100). After 24 hours, the culture medium was removed, and cells were inoculated with E. coli K88 968 (2.52×108 CFU/mL). The percentage of bacteria adhered to the cells was determined using a bacteria enumeration assay. The results showed that MSP Barrier reduced the percentage of adhesion of E. coli K88 968 to the IPEC-1 cell line at the three concentrations tested (-64%, -56% and -50%) compared to the negative control.
Further research has shown that MSP Immunity has the capacity to modulate the synthesis of immune mediators involved in defence mechanisms within the innate and the adaptive immune response, among others, the recruitment and activation of antigen-presenting cells, the differentiation and proliferation of different immune cell populations, while inducing immune tolerance thanks to its anti-inflammatory properties. In vivo scientific studies further confirmed the immunomodulating properties of MSP Immunity, namely by improving the defence activities of monocytes and heterophils in poultry and by favouring the immune transfer through colostrum and milk in swine.
Effect of combined macroalgal extract on gut health
Algimun inclusion in piglets feed during the transition period resulted in a significant reduction of veterinary treatments related to enteric troubles (-57%, P<0.01) and in a reduction of the inflammatory status (-16% haptoglobin level in serum) while improving growth performance (final weight & FCR) leading to a high profitability: ROI = 3:1. In several trials, the use of Algimun in broilers proved to reinforce mucosal barrier function seen by higher concentration of immunoglobulin A in serum and improve immune status of birds as seen by a quick decreased heterophil to lymphocyte ratio over time, always while promoting growth performance.
In short, dietary marine algae extracts can be used as a natural alternative in-feed strategy to strengthen intestinal health by reinforcing its barrier function and by modulating the immune response leading to an improved health and growth performance. This supports on animal resilience represents a good tool to move towards the reduction of antibiotic use.
References available on request.
The For Feed Team can be reached at animalcare.pm@olmix.com.



