Phytate as an anti-nutrient for poultry and swine
Phytate has direct anti-nutritional effects for poultry and swine, causing reductions in performance by lowering amino acid and mineral absorption and increasing endogenous losses. To evaluate this antinutritional effect is not as simple, any change in the diet to reduce phytate concentration will result in other modifications in diet composition that may also interfere with animal performance. One alternative is to use high levels of phytase in the diet, looking for the phytase to hydrolyse and therefore reduce the anti-nutritional effects of phytate.
By Tiago Santos, AB Vista Feed Ingredients, Marlborough, UK
Phytate is a possible source of phosphorous (P) for poultry and swine. Ruminants have a fermentation process where phytate can be broken down by bacteria, releasing P to be absorbed by the animal. However, phytate has long been recognised as a non available P “source” for poultry and swine, as monogastrics do not have enzymes to hydrolyse phytate. Actually, researchers have now shown that the low absorption of phytic P is not related to a lack of ability in poultry and swine to absorb this P, as low mineral diets result in phytic P absorption as high as 70%. Rather, the phytic acid molecule interacts with other minerals and proteins present in the intestinal tract, making phytic P less available (or digestible) for poultry and swine, but also causing an anti-nutritional effect. A better understanding of the effect of phytate on nutrient digestibility and its antinutritional effects opens up an opportunity to improve animal performance, going beyond viewing phytate solely as a possible P source for the animal.
Impact on minerals
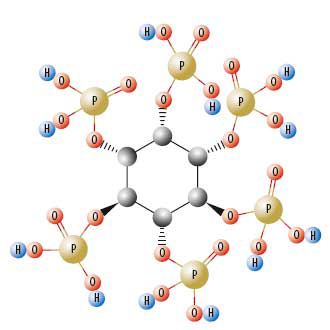
Phytate is a complex molecule with 12 different pKa values (acid dissociation constants), two for each phosphate group. Even at low pH (pH < 2.0) the phytate molecule will still be negatively charged and will become more negative as the pH increases. This is important because when phytate passes through the digestive tract, it goes from a lower pH (in the stomach/gizzard) to neutral pH in the lower intestine. Thus, as pH increases, phytate increases its negative charge and as a consequence, its desire to react with cations (primarily divalent cations such as calcium, zinc and copper). As a result, stable salts are formed which precipitate out of solution (illustrated in Figure 1).
Even though phytate has a higher affinity for cations such as copper and zinc, it is the affinity to calcium that causes majors concerns in animal nutrition due to the higher concentration of this mineral in animal feeds. In vivo trials have already shown that high calcium diets reduce phytic P absorption in broilers, and an increase in phytate concentration in a diet also increases the animal’s requirement for calcium. Once the amount of calcium and phytate in solution exceeds a critical concentration, salt formation and precipitation occurs, reducing the amount of calcium available in the intestine for absorption. The calcium requirement for broilers increases from 0.60% to 0.95% when the phytic P level in the diet increases from 0 to 0.25% – about the phytic P concentration in a standard corn-soybean diet.
In some feeds, primarily piglet prestarter and starter diets, copper and zinc may also be important cations due to high inclusion rates for growth promotion. Blood concentrations can be an indication of animal mineral status and a recent study showed that serum zinc concentrations were reduced in the presence of high phytate, suggesting that high dietary phytate may reduce the availability of zinc and potentially copper (Figure 2). In addition to piglets, animals that have a longer production period, such as turkeys, layers and sows, may also suffer from this interference, causing sub optimal levels of minerals, even though these animals often receive a high level of mineral supplementation.
Impact on protein
Protein digestive processes start in the stomach, where secreted pepsinogen is activated to pepsin, the active endogenous enzyme that initiates hydrolysis of feed protein. In vitro assays have shown that the presence of phytate reduces pepsin activation between pH 0.8 and 2.8. This may result in less protein being initially digested in the acid phase of the digestive process in poultry and swine, and as a consequence impact overall protein digestibility.
Phytate presence also directly reduces protein solubility and consequent digestibility. The initial hypothesis to explain this reduction in digestibility is that at low pH most of the feed proteins, particularly those with high concentrations of basic amino acids, will be well below their isoelectric point and thus positively charged. This would attract the protein molecule to phytate, which is negatively charged at low pH, causing an initial phytate-protein link and consequently a protein-protein link, reducing protein solubility and digestibility.
A new hypothesis proposes that this phytate-protein link does not necessarily occur as high phytate solubility at low pH it would be expected to increase rather than reduce protein solubility. This hypothesis claims that the presence of phytate in solution changes the water conformation of the solution, moving water molecules closer to phytate and further away from the protein molecules. This lower amount of water around the protein molecule would result in a reduction in protein solubility and as a consequence lower digestibility.
Impact on endogenous loss
Phytate can reduce protein solubility and consequent digestibility in the gut. This will result in more undigested protein reaching the duodenum, stimulating gastrin, HCl and pepsinogen secretion in the stomach. We have shown a lower pH in the gizzard at seven and 21 days of age in broilers fed a higher phytate diet, which is presumably related to a higher HCl and pepsinogen production in the proventriculus.
Another consequence of this increase in HCl and pepsinogen production is an increase in endogenous losses by the animal. This is because higher HCl and pepsinogen production has an irritant effect on the gut mucosa, which is compensated by an increased production of mucus as a protective layer. Also, once the digesta gets to the duodenum a greater amount of sodium bicarbonate needs to be secreted by the pancreas to increase pH level and to compensate for the lower stomach pH value. This excess sodium usage can compromise the absorption of amino acids which depend on active transportation via the Na-K pump.
Difficulties in measuring antinutritional effects of phytate
The main problem in studying phytate’s anti-nutritional effect is how to vary its concentration in diets without adjusting other variables (for example fibre levels, mineral digestibility) and determine its effect on animal physiology and performance. One possibility is artificially increasing phytate without significantly changing diet composition, but the source of the phytic acid is very important because of solubility differences as a result of its origin.
Another possibility is to use a higher concentration of phytase in the diets, looking for a quick reduction in phytate concentration in the stomach and thus reducing the anti-nutritional effect. It is important to feed diets not limiting in P to be sure any observed effect is not caused by an increase in P digestibility. This approach is the opposite of that normally recommended when comparing or evaluating phytases.
[Source: Enzymes special]











