The microworld of vitamins and minerals in your premix

Minerals and vitamins can have a huge impact on the quality of feed. The form in which they are provided can have several flow-on effects on the stability of other feed components, such as enzymes, antioxidants and vitamins.
Vitamins are essential nutrients for biological function and serve as co-factors for enzymes involved with energy metabolism. Some of the most notable fat-soluble vitamins added to a premix are vitamin E (cholecalciferol or α-tocopherol acetate are commonly used) and vitamin A (retinol acetate). As they are biological substances, they are sensitive to environmental degradation through exposure to humidity, light, heat and pH.
Degradation of vitamin stability
The addition of trace minerals and vitamins to a premix presents the challenge of negative interactions between the two components, and the impact of trace mineral source is often underestimated. Trace minerals from inorganic sources (especially sulphates) are considered catalysts for free radical formation. Through reduction and oxidation (redox) reactions, the inclusion of mineral sulphates to a premix can lead to rapid degradation of vitamin stability, i.e., the ability of vitamins to retain activity under commercial storage conditions.
As trace minerals can vary in their redox potential (with copper, iron and zinc being the most reactive), vitamins can also vary in their susceptibility to these reactions. Established research has shown that sulphates are particularly reactive, and up to 55% of vitamin B6 activity can be lost in a premix.
Organic minerals
A clear solution is to replace inorganic trace minerals with organic minerals in premixes. Because organic trace minerals are more bioavailable and better utilised by the animal, they can be included at lower inclusion rates.
More and more trace minerals are being manufactured as “organic,” and official terminologies can be vague. This presents an additional challenge when it comes to formulating an optimal premix. Despite “organic trace minerals” being used as a blanket term, the feed industry incorporates a wide range of both complexes and ligands that can vary from single amino acids to hydrolysed proteins, organic acids, and polysaccharide preparations. Additionally, some trace mineral products can have similar — or sometimes worse — efficacy compared to inorganic sulphates and oxides. Hence, it is important to consider not only whether the trace mineral source they are including is organic but whether its biological structure and degree of interaction are effective. This is where data from peer-reviewed, up-to-date publications can help with the decision-making process.
Decrease in vitamin A depends on the source of copper
Recent research examined the effects of 5 commercial copper (Cu) sources (2 proteinates, an amino acid complex, a glycinate and a sulphate) on the antioxidant activity of BHT, the stability of vitamin A and the activity of three phytases in vitro. Although all 5 copper sources showed an inhibitory effect on BHT activity, 3 of the organic copper sources exhibited significantly less antioxidant inhibition than the samples containing the glycinate and copper sulphate (P<0.05). These were 15.6% for proteinate one, 16.3% for proteinate two, 21.9% for the amino acid complex, and 32.1% and 28.9% for the glycinate and sulphate, respectively (Figure 1).
Figure 1 – Impact of copper source on the antioxidant activity of BHT, relative to the positive control.
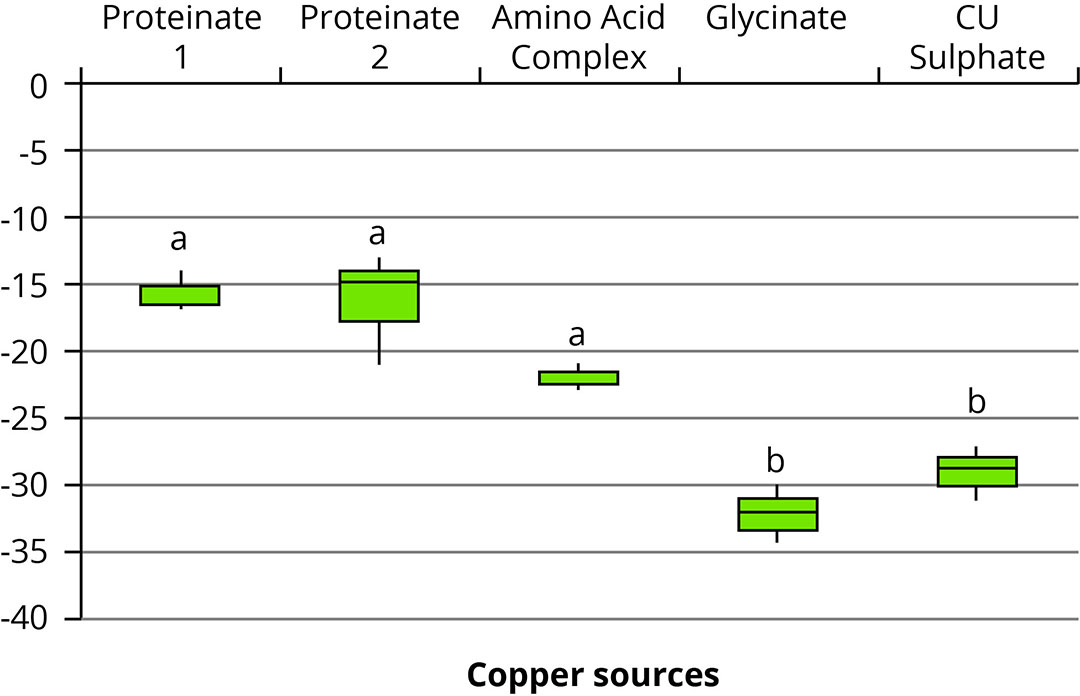
The trial also mirrored commercial conditions where the premixes were stored for a period of 9 weeks. Although the amount of vitamin A decreased across all groups throughout this period, the rate of decrease was found to be dependent on the source of copper present. Proteinate 1 exhibited a loss of only 12.4%, followed by proteinate 2 at 15.7%, both of which did not significantly differ from the vitamin control. The 2 samples that had the highest vitamin A loss were the amino acid complex (25.7%) and the glycinate (31.9%), which were surprisingly higher than the degree of loss in the copper sulphate group (21.1%).
The results also demonstrated that copper had an inhibitory effect on phytase activity and that the rate at which the phytase activity was affected was also dependent on the source and the phytase that was used. In general, proteinate 1 and proteinate 2 led to the lowest rate of phytase activity loss, whereas, in all 3 phytase treatments, they exhibited significantly lower (P<0.05) phytase activity loss than the phytase preparations incubated with Cu sulphate.
Organic proteinate versus an inorganic sulphate
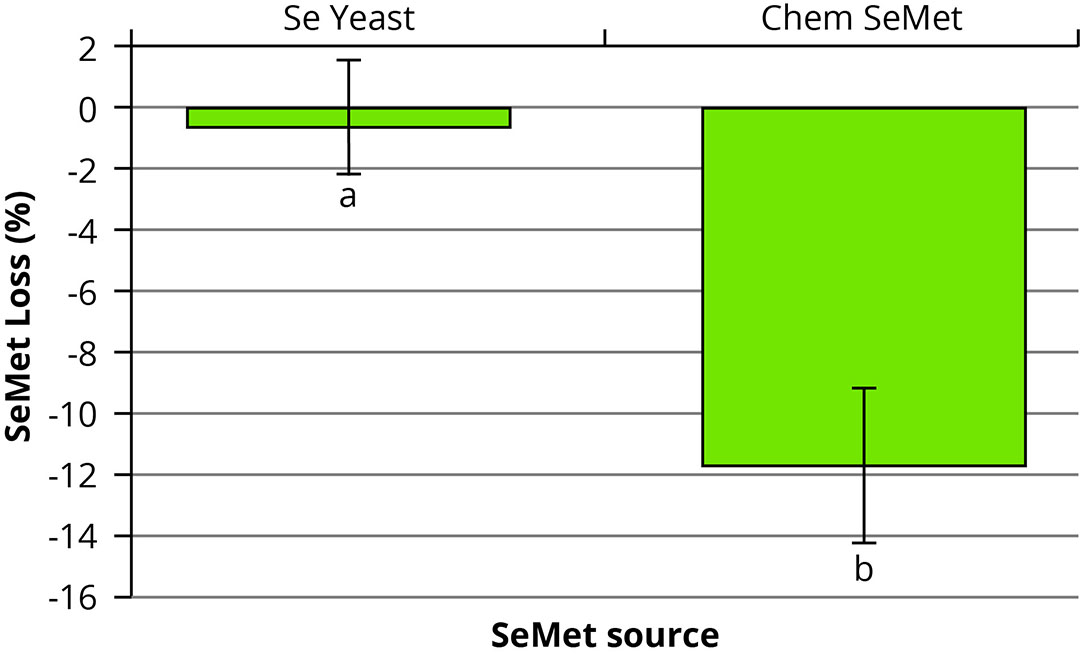
Another recent study compared the effect of an organic proteinate versus an inorganic sulphate mineral source on the stability of vitamin A and E within a premix while also comparing the stability of 2 selenomethionine (SeMet) sources (selenium-enriched yeast and chemically synthesised L-SeMet). It was found that the premixes containing inorganic sulphates had a higher degree of vitamin loss than those formulated with the proteinate trace mineral source. In fact, the inclusion of the trace mineral proteinates significantly reduced inclusion levels (P<0.05) while increasing the stability of both vitamins when compared to the inorganic dietary treatments. The SeMet content was analysed after 49 days in storage using liquid chromatography, which showed that the SeMet present in the SeYeast source was significantly more stable (P<0.05) compared to the chemically synthesised L-SeMet (Figure 2).
Figure 2 – Selenomethionine (SeMet) loss between two SeMet sources in the presence of NRC-recommended inclusion rates of vitamins and trace minerals after a seven-week storage period. SeYeast = selenium-enriched yeast; Chem SeMet = chemically synthesised L-selenomethionine.
Conclusion
A premix contains a ‘microscopic economy’ of feed components that, based on recent peer-reviewed research, must be carefully balanced when interactions are considered. Data published in 2021 has shown that even though trace minerals and vitamins do interact within a premix, the degree of interaction is strongly dependent on the trace mineral source, with organic trace mineral proteinates having a significantly lower inhibitory effect compared to other sources.
References for this article are available upon request.











