Mycotoxins – a rising threat to aquaculture
Plant-based ingredients are increasingly used in fish diets due to increase economic/market pressures on feed compounders to produce lower cost, sustainable alternatives. Because plant ingredients pose a high risk of mycotoxin contamination, moving to plant protein sources in the aquafeed industry demands careful risk assessment, as well as appropriate protection strategies for fish fed contaminated feeds.
| *)Peter Spring lectures at the Swiss College of Agriculture, Zollikofen, Switzerland and Daniel Fegan works for Alltech Inc., Bangkok, Thailand. Spring presented an extended version of this paper at Alltech’s annual conference in Lexington, Kentucky. |
In omnivorous fish, such as channel catfish (lctalurus punctatus), nutritional formulation has developed over the last decade to include little or no animal protein. Because plant ingredients pose a high risk of mycotoxin contamination, moving to plant protein sources in the aquafeed industry demands careful risk assessment regarding mycotoxins, as well as the development of appropriate protection strategies for fish fed contaminated feeds.
Mycotoxins are naturally occurring, toxic chemical compounds produced by filamentous fungi. These moulds can infect agricultural crops, particularly cereals and oilseeds, during crop growth, harvest, storage or processing. If the conditions for fungal growth and metabolism are right, mycotoxin contamination is often the result. Thus, production of toxic metabolites can occur during the growth of the crop, during post-harvest storage or during the storage of the compounded feed.
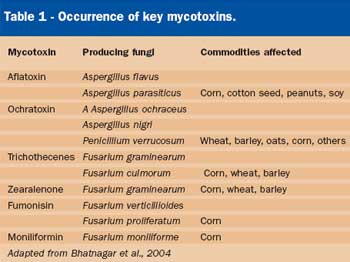
The use of more plant-based ingredients in aquafeeds enhances both the risk of introducing mycotoxins into the feed at the point of feed manufacturing, and mycotoxin production during storage of compounded feed. Through contamination with fungal spores, plant ingredients can infect the final diet with spores. If temperature and moisture in the storage environment allow for fungal growth, additional mycotoxins can be produced in between manufacturing and use of the feed. Since conditions for fungal growth vary greatly between the field and storage, different fungal populations may result, producing cocktails of mycotoxins. This possibility must be considered when conducting a risk assessment and implementing preventive measures. Although several hundred mycotoxins are known, the mycotoxins of most concern, based on their toxicity and common occurrence, are aflatoxin, ochratoxin A, the trichothecenes (DON, T-2 toxin), zearalenone, fumonisin, and moniliformin (Table 1).
Mycotoxicoses
Mycotoxins are structurally very diverse, a characteristic that leads to a wide range of symptoms in mycotoxin affected animals. The mode of action of mycotoxins is grouped by three primary mechanisms:
Alteration in the content, absorption and metabolism of nutrients;
Changes in endocrine and neuroendocrine function; and, most importantly, Suppression of the immune system.
The effects on the immune system are of particular importance as they predispose animals to infectious diseases and reduce productivity. The fact that most of the symptoms of mycotoxicoses are rather nonspecific and can have multiple causes often makes it difficult to properly diagnose mycotoxin problems. General symptoms (reductions in performance and immune status) are seen when dealing with moderate toxin levels, while symptoms caused by higher toxin levels are more specific.
Further complications in mycotoxicosis diagnoses can be caused by secondary symptoms resulting from opportunistic disease related to the suppression of the immune system after mycotoxin exposure. To effectively recognise mycotoxicosis, experience with mycotoxin-affected animals is important. This experience, combined with adequate feed and tissue analyses, provides the most accurate diagnosis of mycotoxicosis.
Aflatoxin
Aflatoxin is produced primarily by Aspergillus flavus, and is a major concern due to its carcinogenicity and ubiquity, especially in warm and humid climates. It can be produced both in the field and during storage. Due to the growth requirements of the fungi, aflatoxin poses a particular risk in warmer climates. Since aflatoxin is transferred at low rates into edible tissues, it is not only of concern for animal health, but also for the health of humans consuming food of animal origin. Therefore, in some countries the regulatory authorities have set upper limits for aflatoxin in feeds and animal products. In these markets, ingredients or diets with aflatoxins that exceed these limits must be removed and destroyed.
Aflatoxin was the first of the mycotoxins to be investigated in aquaculture. As in other animal species, aflatoxin exerts carcinogenic effects in fish. Different research groups have reported that long-term exposure of <1 ppb of dietary aflatoxin B1 can be sufficient to cause hepatomes.
The complexity of the task has made it impossible to determine a safe level for mycotoxins in feeds. Adverse effects depend not only on the dietary concentration, but also on the length of exposure, the fish species and the age of the fish, their nutritional status and health status.
Since aflatoxin can impair immune function exposure increases fish susceptibility to disease. A healthy fish is less likely to succumb to secondary infections and has a greater tolerance for the toxin.
In general it can be concluded that aflatoxin B1 concentrations commonly found in feed are in the range that significantly impair aquatic animal health and performance. Therefore, measures to reduce exposure must be taken when striving for maximum health, performance and economic results.
Other toxins
Ochratoxin A has not been studied to the same extent as aflatoxin in aquaculture. Ochratoxin can be present in cereal grains and oilseeds, and is often formed during ingredient or diet storage. Ochratoxin A is primarily produced at higher temperatures by Penicillium verrucosum. Thus, like aflatoxin, ochratoxin is more often found in warmer climates. The key target organ of ochratoxin is the kidney, where it causes necrotic lesions in the proximal tubules.
Trichothecenes and zearalenone are produced in temperate climates by the moulds Fusarium graminearum and Fusarium culmorum. These toxins are produced in the field and enter fish diets as grain contaminants; and they continue to be produced during storage. Zearalenone has oestrogen-like activity that has detrimental effects on the fertility of mammals, although it is probably of less importance in aquaculture.
Trichothecenes have been intensively researched, and are known to affect aquatic species. Chemically, they are structurally similar to compounds such as deoxynivalenone (DON), T2-toxin and diacetoxyscirpenol (DAS). In mammalian and avian species, trichothecenes have been shown to reduce feed intake and performance and impair immune function. Diets containing DON reduced live weight gain in juvenile rainbow trout, as a result of poor feed intake and feed conversion. Catfish, however, seem more able to tolerate dietary DON ,but seem quite susceptible to T-2-toxin.
In shrimp DON concentrations as low as 0.2 ppm led to significant reductions in growth rate. Toxic levels reported in both trout and shrimp seem comparable to concentrations reported in swine, where concentrations of 1 ppm or more are considered problematic, although in young piglets, lower concentrations have been shown to reduce feed intake.
Fumonisin is of concern to the aquaculture industry because it commonly contaminates corn and its by-products. Over 25 countries in the world have reported natural occurrence of fumonisin in feed and foodstuffs. Young fish are more susceptible to the toxin than older fish. Fumonisin also effects on the immune system. However, because no quick analytical test for this biomarker is available today it is rarely searched for in field situations.
It is know that moniliformin reduces weight gain in catfish. When feeding moniliformin in combination with fumonisin, a synergistic negative interaction between the two toxins on weight gain was observed. This is in agreement with research data from mammalian and avian species. Since toxins are often present as a cocktail in a single ingredient or a final diet, toxic effects due to synergistic action occur on a daily basis in the field. One has, therefore, to be careful when interpreting mycotoxin concentrations and potential risks. A concentration that does not adversely affect animal performance in a semi-purified diet can lead to problems in a natural diet in the presence of a mycotoxin cocktail.
Toxin absorption
One of the most effective methods of reducing the effects of mycotoxins is the inclusion of a mycotoxin adsorption agent. This corrective action can only be taken if the mycotoxin concentrations are below legal limits (e.g., aflatoxin legal limits for raw materials in certain markets). An effective sequestering agent is one that tightly binds mycotoxins in contaminated feed without disassociating from them in the gastrointestinal tract of the animal. The toxin-sequestrant complex can then pass safely through the animal and be eliminated via the faeces, minimising animal exposure to mycotoxins.
The following guidelines should be utilized when evaluating a mycotoxin binder:
- High level of specificity and affinity for a wide range of different mycotoxins
- No absorption of minerals, vitamins and drugs
- Low level of inclusion
- Quality control (no contaminants)
- Stability over different pH values
- Scientifically tested in controlled in-vitro and in-vivo studies
Conclusions
Research comparing mycotoxins in different aquatic species has demonstrated that they pose a risk to fish and shrimp performance and health. The exposure to mycotoxins increases with heavier reliance on plant-based raw materials, since these ingredients pose a higher risk of mycotoxin contamination than animal-based products. With increased use of plant ingredients, mycotoxin risk assessment plans, as well as the appropriate prevention strategies, should be put in place in any aquatic production system.
Prevention strategies must target the production chain from cropping systems to animal feeding. Adsorbents that bind mycotoxins and decrease their bioavailability show a great deal of promise in strategies that attenuate mycotoxin-induced toxicosis.











