Fumonisins – Underestimated but controllable by biotransformation
Fumonisins are frequently occurring mycotoxins with great relevance in animal feed. An effective way to counteract them is through biodegradation by natural metabolic pathways, a process called biotransformation. In contrast to adsorption of mycotoxins by clays, microbial and/or enzymatic biotransformation is highly specific and irreversible.
By Verena Starkl and Karin Nährer, Biomin, Austria
In 1989, the US corn crop was highly contaminated by fumonisins resulting in large outbreaks of equine leukoencephalomalacia (ELEM) and porcine pulmonary edema (PPE) among swine. Subsequently, fumonisins (FUM) were recognised as a worldwide contaminant of not only animal feed but also food and especially corn. In a recent publication FUM were found to be the most frequently occurring mycotoxins worldwide (64% of all samples investigated) in the different commodities investigated. Reviewing the occurrence of mycotoxins in DDGS samples, fumonisins were found in 91% of DDGS samples and an average contamination of 1,036 μg/kg was reported.
Fumonisins are all over
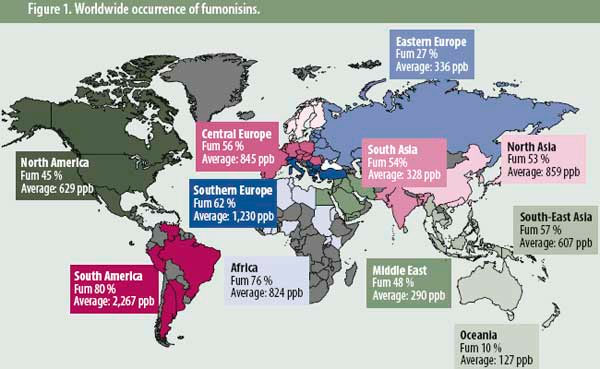
To show the importance of fumonisin occurrences, Biomin conducted a worldwide mycotoxin survey. The data focuses on results obtained between 2004 and 2011, in which more than 9,000 samples (see Figure 1) were collected worldwide and analysed for fumonisins. The fumonisins recorded consisted of fumonisin B1 and fumonisin B2. Analyses were performed either by high performance liquid chromatography or enzyme-linked immunosorbent assay according to standard procedures. For the purpose of data analysis, nondetection levels were based on the quantification limits of the test method.
Several fungi are known to produce fumonisins in different quantities under similar conditions. The most common producers of fumonisins are Fusarium verticillioides (former F. moniliforme) and Fusarium proliferatum which are also the most common fungi associated with maize.
The chemical structure of fumonisin was first reported in 1988. Since then, more than 28 homologues have been discovered and more are likely to be found. FB1 is the most common, and from a toxicological standpoint, the most thoroughly studied.
How do fumonisins affect animals?
Fumonisins are known to inhibit the biosynthesis of sphingolipids, induce hepatotoxicity, possess renal toxicity and elevate serum cholesterol concentrations in all studied species. Since their discovery in the late eighties in South Africa, FUM have been connected to a series of diseases which are tremendously speciesspecific in terms of pathology. These diseases include oesophageal cancer in certain human populations as well as leukoencephalomalacia or “hole-in-thehead- disease” in horses. In swine, fumonisin contamination is associated with porcine pulmonary edema. This seems to be due to the capacity of FB1 to decrease cardiac contractility, mean systemic arterial pressure, heart rate and cardiac output, and increased mean pulmonary artery pressure and pulmonary artery wedge pressure. Therefore, fumonisin-induced pulmonary edema apparently results from acute left-side heart failure and increase in tissue sphingosine concentrations which have an impact on cardiac contractility.
Furthermore, this also leads to decreased heart rate. As the contractile rate of the heart decreases, mean arterial pressure falls below the level needed for the regulation of the cardiac circulation and pulmonary oedema forms.
Negative impact on vaccination results
In addition, FUM show a broad impact on the immune system. Chronic exposure to fumonisin B1 decreased the proliferation of undifferentiated porcine epithelial intestinal cells, altered the integrity of intestinal epithelium and consequently facilitated the intrusion of pathogens into the body. A 28-day administration of 8 mg fumonisin B1/kg feed to weaning piglets caused a significant decrease in antibody titer after vaccination for Mycoplasma agalactiae. Fumonisin-induced impaired vaccination responses also reduced the level of specific antibodies and the period of vaccine protection. Such effects are very often undetected as they are asymptomatic, but they may cause substantial economic losses. In poultry, fumonisins reduce thymus weight, decrease antibody response to sheep red blood cells and immunisation against Brucella abortus and Newcastle disease. FUM also reduce macrophage number, phagocytic ability of macrophages and decrease total white blood cell counts in poultry. The impact of fumonisins on the immune system may apparently be more silent and therefore more easily forgotten – however it always has to be considered.
Under field conditions animals are exposed to different mycotoxins since most of the common fungi can produce more than one type of mycotoxins. Additive and, more precarious, synergistic interactions have been described between fumonisins and other major mycotoxins, which lead to more severe effects in animals. Besides aflatoxins and ochratoxins, combined effects were also investigated with deoxynivalenol, the most frequently detected fusariotoxin. Data indicate that the ingestion of diets containing FUM and DON together induces more histopathological lesions and greater immune suppression.
Distribution and carry-over
Fumonisins in general, under natural conditions, only accumulate insignificantly in the tissues. The most important target organs are the liver and the kidneys depending on species and dosage. In pigs orally dosed with radiolabelled (14C) FB1, the 14C is distributed to most tissues, with liver and kidney containing the highest concentration of radio labelled C. It was estimated that exposure to dietary FB1 at 2-3 mg/kg in feed would require a withdrawal period of at least two weeks for the fumonisin to be eliminated from the liver and kidneys. In chickens and dairy cows the poor absorption (<1%) was reflected by the fact that only trace amounts of radioactivity were recovered in tissues, no residues were recovered in eggs of laying hens, and no fb1 or hydrolysis products (hfb1) were recovered in milk.>
Counteraction
After years of scientific investigation Biomin discovered Fumzyme, an enzyme that degrades fumonisins to non-toxic hydrolysed FB1 (HFB1). A recently published study describes that HFB1 does not cause intestinal or hepatic toxicity in a very sensitive pig model and does not induce major changes in the sphingolipid metabolism. Consequently, this suggests that the enzymatic conversion of FB1 into HFB1 is a feasible strategy to counteract FB1 exposure.
A trial was conducted at the Samitec Institute (Brazil) in cooperation with Lamic, Universidade Federal de Santa Maria (Brazil), with the aim to evaluate the efficacy of Mycofix® Focus, a product including the fumonisin-degrading enzyme Fumzyme, in diets of weaning piglets contaminated with fumonisin B1. The sphinganine/sphingosine ratio, a biomarker that is scientifically recognised to indicate fumonisin contamination, was significantly increased in the group where 50 ppm of fumonisin were fed, and was significantly reduced where Fumzyme was added to degrade fumonisins in the diet. In vivo efficacy of the enzyme was also confirmed by improving the relative liver weight (Figure 2).
This leads to the conclusion that an effective way to counteract mycotoxins is through biodegradation by natural metabolic pathways, a process called biotransformation. This microbial and/ or enzymatic biotransformation, in contrast to mycotoxin adsorption by clay, is highly specific and irreversible.
References available on request.
[Source: Mycotoxin Special]











